To conduct electrophoresis of single-cell lysate, an external electric field is applied across cell-laden microwells (fabricated in agarose or polyacrylamide) to initiate and drive electrophoretic separation. Gravity is commonly used to sediment single cells into open microwells, followed by in situ cytometry. Microwell arrays have been used for high throughput and automatic analysis of DNA and protein from hundreds to thousands of cells. Microwells are widely utilized to isolate intact single cells for subsequent analysis. 
Such protein markers to assess technical variation in electromigration would be invaluable for quantifying proteoform expression that drives drug resistance in single cancer cells. Run-to-run reproducibility in electromigration is monitored using protein markers, providing a quality control indicator. In western blotting, protein markers not only estimate the molecular mass of a target, but also indicate the quality of protein transfer from the PAGE slab gel to the immunoblotting membrane. In slab-gel protein polyacrylamide gel electrophoresis (PAGE), the first or last separation lanes of the slab gel are used for analysis of protein markers. Consequently, protein markers and ladders find utility in slab-gel, capillary, and microchannel electrophoresis formats. Well-validated protein standards are also crucial in identifying and understanding “gel shifting,” a phenomenon in which a protein does not electromigrate proportionally with its molecular mass due to amino acid substitutions, post-translational modifications, or other unknown factors. In order to accurately size and identify targets, protein markers control for technical variation in the uniformity of the electric field, sieving matrix structure, and buffer composition-all of which influence protein electromigration. Īnalysis of cell lysate by electrophoretic separations uses protein markers to estimate the molecular mass of a target from the known masses of the markers. In microfluidic protein assays of a single cell lysate (e.g., single-cell barcode assay), estimates of technical variation are inferred from multiple measurements of each lysate. In flow cytometry, lasers are calibrated before cell sorting using fluorescent microparticles. In single-cell sequencing, synthetic spike-ins and unique molecular identifiers (e.g., short-random DNA sequences) directly measure error rates, analytical sensitivity, and biases stemming from sample preparation. Molecular standards play an important role in deconvolving biological variation from technical variation arising from a measurement tool. Single-cell analysis tools report biomolecular heterogeneity that drives processes from immune-cell response to cancer progression. Microparticle-delivered protein standards underpin robust, reproducible electrophoretic cytometry that complements single-cell genomics and transcriptomics. The protein marker and microparticle system is applied to determine the molecular masses of five endogenous proteins in breast cancer cells (GAPDH, β-TUB, CK8, STAT3, ER- α), with <20% mass error. 
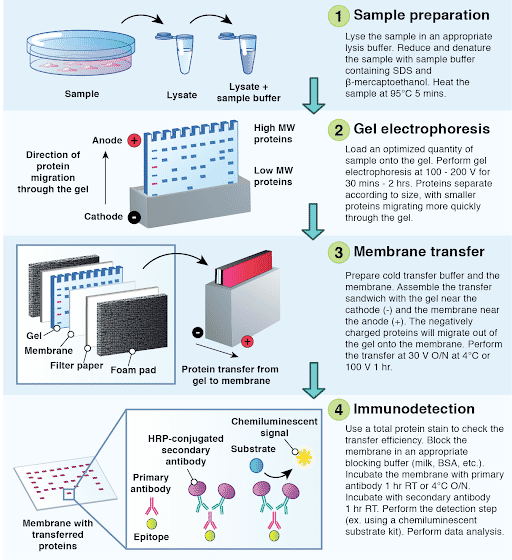
Across hundreds of individual microwells and different microdevices, robust log-linear regression fits ( R 2 > 0.97) of protein molecular mass and electrophoretic mobility are observed. Imidazole in the cell lysis buffer solubilizes protein markers during a 30 s cell lysis step, with an observed protein marker release half-life of 4.46 s. Nickel-coated microparticles are designed, fabricated, and characterized, each conjugated with a mixture of histidine-tagged proteins (42.3–100 kDa). A magnetic field directs protein-coated microparticles to >75% of microwells, so as to 1) deliver a quantum of protein marker to each cell-laden microwell and 2) synchronize protein marker solubilization with cell lysis. To incorporate protein markers in single-cell western blotting, microwells are used to isolate individual cells and protein marker-coated microparticles. To accurately size protein targets, sample analysis includes concurrent analysis of protein markers with known molecular masses. Immunoblotting confers protein identification specificity beyond that of immunoassays by prepending protein electrophoresis (sizing) to immunoprobing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
